 It is affected by its atomic number and the size of the atom. ElectronegativityĮlectronegativity is the tendency for an element to attract electrons towards itself. In ionic bonding, the compound's net charge must be zero, with the same amount of electrons being gained and lost. By losing an electron or two, the metals can satisfy the octet rule and can have a complete valence shell, while nonmetals can accept electrons to fulfill the rule as well. The metal loses electrons to become a positively charged cation, while the nonmetal gains electrons to become a negatively charged anion. Ionic bonding is the complete transfer of valence electrons between a metal and a nonmetal. Therefore, it tends to share covalent bonds with other elements instead! Ionic Bonds The most common element this occurs with is Carbon (C): carbon has four valence electrons, and it is highly unfavorable for it to either lose or gain four more electrons to satisfy the octet rule. These can be either single bonds (where one valence electron is shared), double bonds (where two valence electrons are shared), or triple bonds (where three valence electrons are shared). This type of bonding can usually be observed between nonmetals or atoms close to each other on the periodic table, with similar electronegativities (explained further below!). In a nutshell, a covalent bond is when two atoms share a valence electron and form a bond. However, because none of the atoms have complete valence shells except for the noble gases (the left-most column on the periodic table), they all tend to bond together to complete each other's valence shells. BondingĮvery atom's goal is to have a complete valence shell with 8 electrons (or, in Hydrogen's case, 2). In this way, you can predict □ which elements will bond with each other based on how many more or fewer electrons the atoms need to have a complete valence shell. 
The number of valence electrons an atom has can tell you a lot about its reactivity.Ītoms with one or two extra valence electrons that form a complete shell (known as the alkali and alkaline earth metals, respectively) are highly reactive, and so are those with one or two less than a full shell (groups 17 (halogens) and 16). They vary quite unpredictably, and therefore we can't predict ❌ the number of valence electrons for transition elements. So why don't transition elements work like every other element? The answer is long and complex but boils down to orbitals- the paths electrons take around the nucleus. 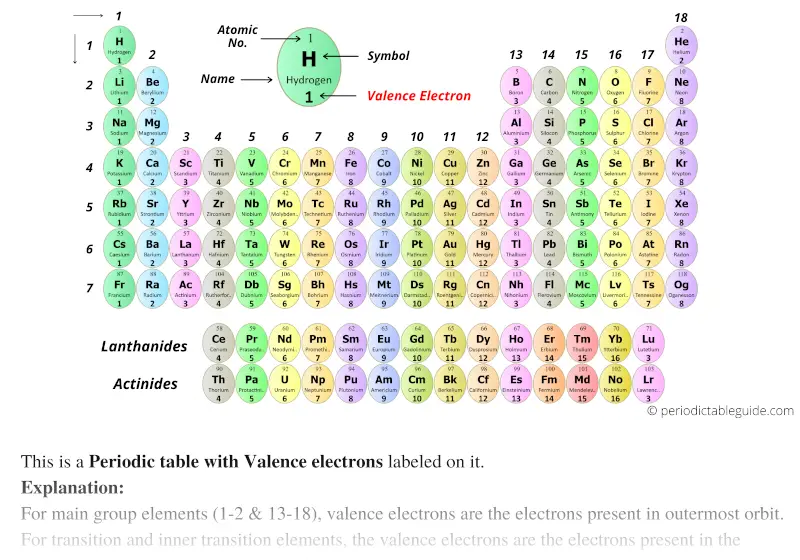
The rules mentioned above don't apply to the transition and inner transition elements (another name to refer to lanthanides and actinides)! However, these rules only apply to main group elements in Groups 1-2 and 13-18. □ To figure out how many electron shells an atom has encircling its nucleus, just look at the period number! Look at the unit digit of the group number to determine the number of valence electrons for elements in that group.  
When you look across a period, the number of electron shells stays the same, but the number of valence electrons increases incrementally. For example, both Beryllium (Be) and Calcium (Ca) have two valence electrons, but Beryllium has 2 electron shells while Calcium has 4. When you look down a group, the number of valence electrons of an element remains the same, but the number of shells increases. The transition elements form a bridge between Groups 2 and 13, with the lanthanides and actinides included. The table contains 18 columns (known as groups) and seven rows (known as periods). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
